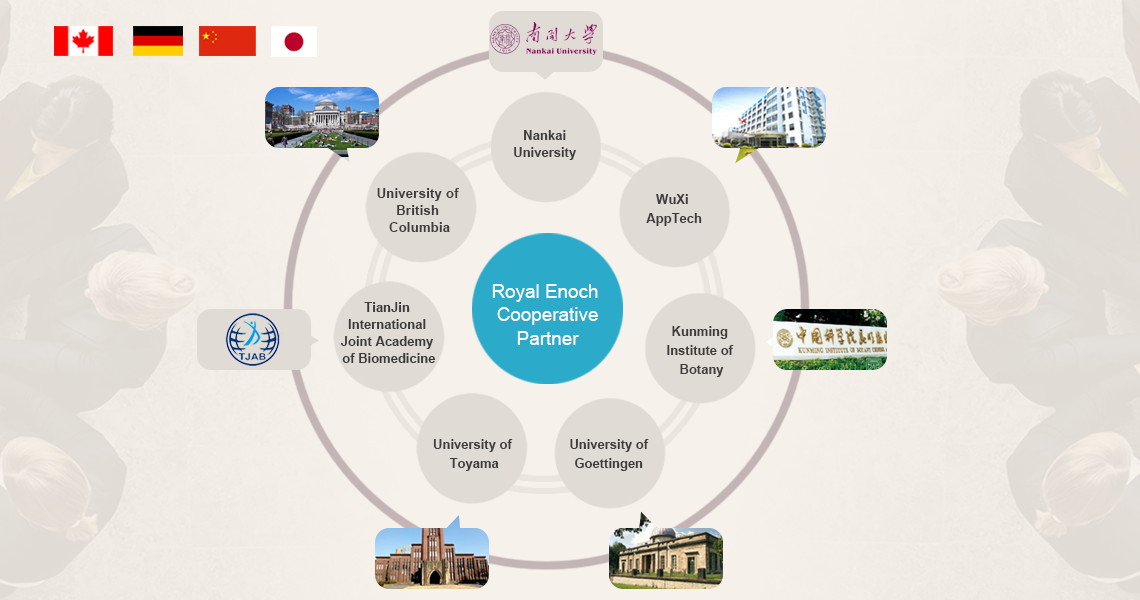
Canada Royal Enoch Phytomedicine Ltd.
Our headquarter is located in Vancouver, Canada. We are a hi-tech enterprise group mainly engaged in the research, production, and sales of natural plant-based products. We aim to extract active ingredients from natural herbal plants.
Development Timeline

-
2005Began to produce highly-purified rare ginsenosides Rg3, Rh2, Rh1, aPPD and aPPT etc., in large scale, at the same time, successfully developed natural plant skin care products with anti-oxidant, anti-aging, and skin-tone-evening properties. These products were sold in beauty salons.
-
2009Developed the rare ginsenosideaglycone protopanaxadiol (aPPD) micro packaging soft gelatin capsules.
-
2010Achieved a break-through in our proprietary technology production of the highly purified aglycone protopanaxadiol (aPPD) and the aglycone proto- panaxatriol (aPPT) by oxidation catalysis.
-
2011Developed an industrialized production technology in transforming protopanaxadiol to rare ginsenosides Rk1, Rg5, Rk2, Rh3, at the same time, successfully developed water-soluble and lipid-soluble gelatin capsules.
-
2012North American and European users began to take the Redsenol-1 Noble Ginsenosides capsules.
-
2013-2015Tested rare ginsenosides'effects through in vitro cell experiments, and obtained confirming results on their broad-spectrum activity.
-
2016Worked on the market promotion of the Redsenol-1 Noble Ginsenosides capsules.
-
2017Meticulously selected eight highly active rare ginsenosides for the preparation-----Redsenol-DAG sublingual pills.
Future Directions
Enterprise Culture



A Great Scientific Achievement
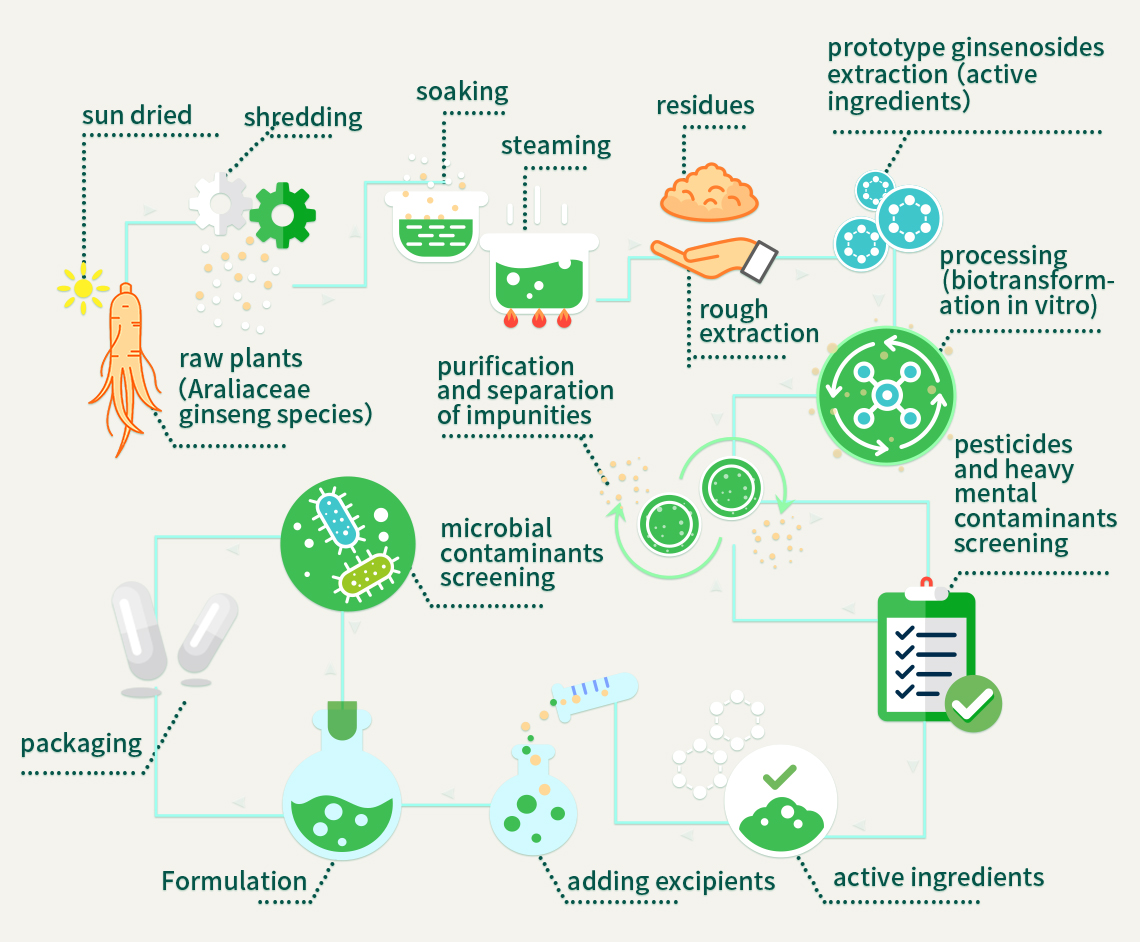
Canada Royal Enoch Phytomedicine Ltd. selects high-quality Araliaceae ginseng species (Asian ginseng, American ginseng, panax notoginseng, etc.) and transforms them into 16 rare ginsenosides, namely Rk2, Rg3(S, R), Rh2(S, R), Rg5, Rk1, Rk3, Rh1(S, R), Rh3, Rh4, aPPT(S, R), aPPD(S, R), on a large scale with propriety technology. These sixteen rare ginsenosides are further prepared and formulated to Redsenol capsules. Eight highly active rare ginsenosides are meticulously selected to formulate Redsenol-DAG sublingual pills. Redsenol meets U.S. Pharmacopeial Convention (USP), each capsule has gone through 70 strict screenings by Labs-Mart Inc. located in Canada (i.e. 5 heavy metal contaminants screenings, 5 microbial contaminants screenings, 60 pesticide residue screenings). Redsenol Rare Ginsenosides capsule is confirmed to be safe and effective, suitable for long-term use.
R&D Milestones

Phase 1: 2005—2010
The R&D team conquered technical barriers in bulk production of the rare ginsenosides Rg3,Rh2,Rh1, aPPD,aPPT,etc..

Phase 2: 2010—2012
The R&D team conquered technical barriers in industrialized production of the more hard-to-obtain rare ginsenosides Rk1, Rk2, Rh3, Rg5, etc. We are now the first institution in the world possessing such technology.

Phase 3: 2012—2014
The R&D team conducted plenty of research in rare ginsenosides Rk1, Rk2, Rh3, Rg5,etc., and the results confirmed their roles in enhancing human health.
Standard Production Process
Standard Manufacturing Workshop

Core R&D Team

Lead by the Chinese Canadian scientist Peihua Yu PhD.
Devoted in phytomedicine research on Araliaceae ginseng species (Asian ginseng, American ginseng,
panax notoginseng, etc.), for more than 10 years,
the team developed its own proprietary technology
that successfully conquered technical barriers in transforming prototype ginsenosides in industrialized
settings,
thus yielding readily-absorbable and effective rare ginsenosides.
Dan Deng
Chemical engineer
Canada Royal Enoch Phytomedicine Ltd. chief chemical engineer
Martin Wang
Ph.D. in Chinese medicine
Ph.D. in Chinese medicine, Nanjing University of Chinese Medicine Post-doctoral fellow, Toyama Medical and Pharmaceutical University and Chinese Medicine Research Institute
Muyuan (Sophie) Zhong
Msc in Pharmacology and Biomedical Toxicology
Msc in Pharmacology and Biomedical Toxicology, University of Toronto.Core member of the R & D team